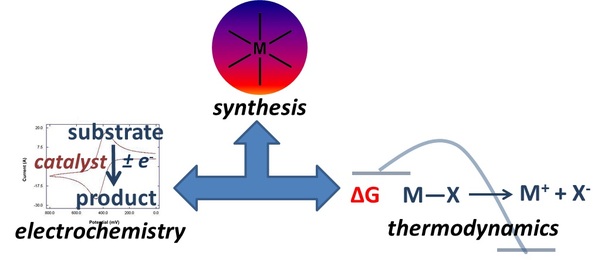
The Yang group is focused on the development of catalysts for the production and utilization of chemical fuels. In most cases, an applied electrochemical potential will be used to drive endothermic reactions to generate chemical fuels. The initial targets will include the reduction of water to hydrogen and carbon dioxide to more energy-dense C1 products. We are also interested in developing efficient and fast electrocatalysts for the corresponding reverse reactions, which is necessary for chemical fuel utilization in fuel cells.
The laboratory research involves the synthesis and characterization of inorganic coordination compounds. An important component in this research is the quantification of bond strengths in transition metal complexes to determine the overall free energy of the target reactions.This information is critical for optimization of intermediate reaction energies in catalytic cycles.
Since most of the reactions of interest involve oxidations and/or reductions, electrochemical studies will play a large role in studying the complexes and evaluating their function as electrocatalysts. Detailed mechanistic and kinetic studies will complement the electrochemical studies to achieve insights into improving catalyst design.
The laboratory research involves the synthesis and characterization of inorganic coordination compounds. An important component in this research is the quantification of bond strengths in transition metal complexes to determine the overall free energy of the target reactions.This information is critical for optimization of intermediate reaction energies in catalytic cycles.
Since most of the reactions of interest involve oxidations and/or reductions, electrochemical studies will play a large role in studying the complexes and evaluating their function as electrocatalysts. Detailed mechanistic and kinetic studies will complement the electrochemical studies to achieve insights into improving catalyst design.